|
Variations in N 2O 5 reactivity in aerosols can result in significant losses in tropospheric ozone, hydroxyl radicals, and NO x concentrations. Dinitrogen pentoxide has also been observed in the stratosphere at similar levels, the reservoir formation having been postulated in considering the puzzling observations of a sudden drop in stratospheric NO 2 levels above 50 °N, the so-called ' Noxon cliff'. Mixing ratios of several parts per billion by volume have been observed in polluted regions of the nighttime troposphere. In the atmosphere, dinitrogen pentoxide is an important reservoir of the NO x species that are responsible for ozone depletion: its formation provides a null cycle with which NO and NO 2 are temporarily held in an unreactive state. This salt retains the high reactivity of NO + 2, but it is thermally stable, decomposing at about 180 ☌ (into NO 2F and BF 3).ĭinitrogen pentoxide is relevant to the preparation of explosives. In this use, N 2O 5 has been largely replaced by nitronium tetrafluoroborate + −. The reactivity of the NO + 2 can be further enhanced with strong acids that generate the "super- electrophile" HNO 2+ 2. This nitration reaction is represented as follows: Applications Nitration of organic compounds ĭinitrogen pentoxide, for example as a solution in chloroform, has been used as a reagent to introduce the −NO 2 functionality in organic compounds. The rate of the initial reaction between dinitrogen pentoxide and nitric oxide of the elementary unimolecular decomposition. N 2O 5 can also be decomposed in the presence of nitric oxide NO: Ln P = 23.2348 − 7098.2 T Decomposition of nitrogen pentoxide in the presence of nitric oxide The vapor pressure P (in atm) as a function of temperature T (in kelvin), in the range 211 to 305 K (−62 to 32 ☌), is well approximated by the formula It crystallizes in the space group D 4Ħ h ( C6/ mmc) with Z = 2, with the NO − 3 anions in the D 3 h sites and the NO + 2 cations in D 3 d sites. Both nitrogen centers have oxidation state +5. Pure solid N 2O 5 is a salt, consisting of separated linear nitronium ions NO + 2 and planar trigonal nitrate anions NO − 3. N 2O 5 was first reported by Deville in 1840, who prepared it by treating silver nitrate ( AgNO 3) with chlorine. The solid is a salt, nitronium nitrate, consisting of separate nitronium cations + and nitrate anions − but in the gas phase and under some other conditions it is a covalently-bound molecule. N 2O 5 is a rare example of a compound that adopts two structures depending on the conditions. ĭinitrogen pentoxide is an unstable and potentially dangerous oxidizer that once was used as a reagent when dissolved in chloroform for nitrations but has largely been superseded by nitronium tetrafluoroborate ( NO 2BF 4). It exists as colourless crystals that sublime slightly above room temperature, yielding a colorless gas. It is one of the binary nitrogen oxides, a family of compounds that only contain nitrogen and oxygen. To determine if a molecule has a permanent dipole moment (and hence experiences dipole-dipole forces) you need to be able to draw a Lewis electron-dot structure for the molecule and apply VSEPR theory to determine the shape of the molecule.Dinitrogen pentoxide (also known as nitrogen pentoxide or nitric anhydride) is the chemical compound with the formula N 2O 5. 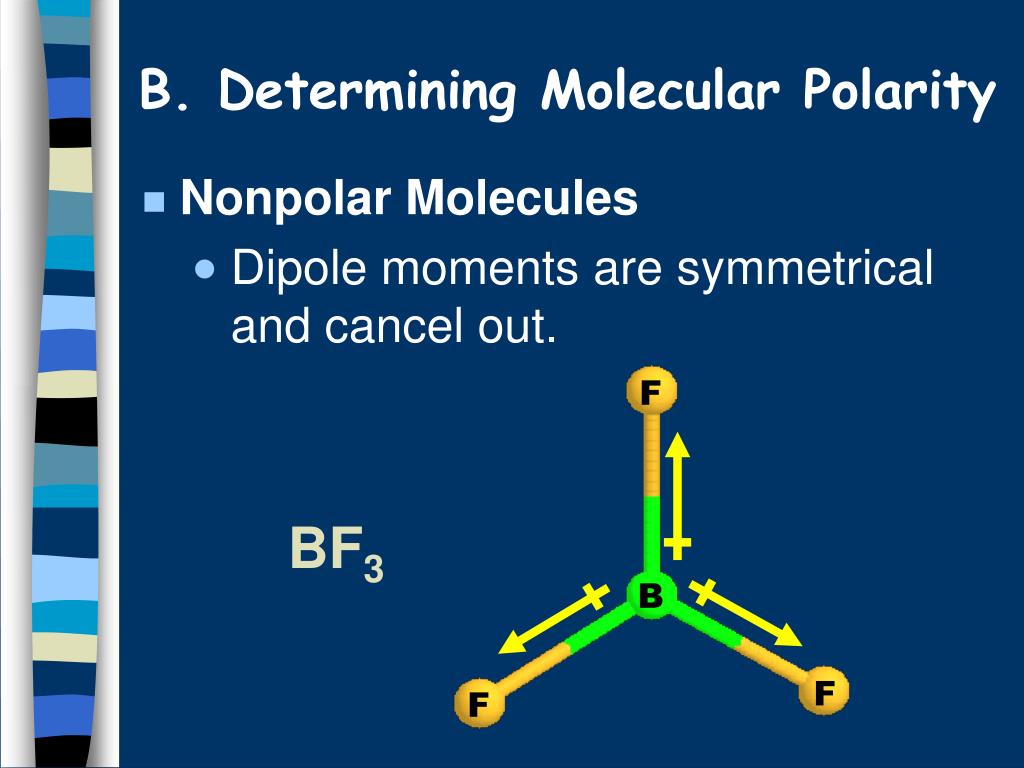
For example, in the molecule BF 3, each BF bond has a dipole moment with a partial negative charge on the fluorine (it is much more electronegative than the boron), but the shape of the molecule (trigonal planar) is such that the bond dipole moments add up to zero. These bond dipole moments will be large if the bonded atoms have very different electronegativities (see this WebElements page for a graph of electronegativity across the periodic table). You can think of each chemical bond in a molecule as having a dipole moment oriented parallel to the bond, with a size and direction determined by the atoms at each end of the bond. So it is the shape of a molecule that is important in determining whether it has a dipole moment.Ī dipole moment is a vector quantity - it has both size and direction. 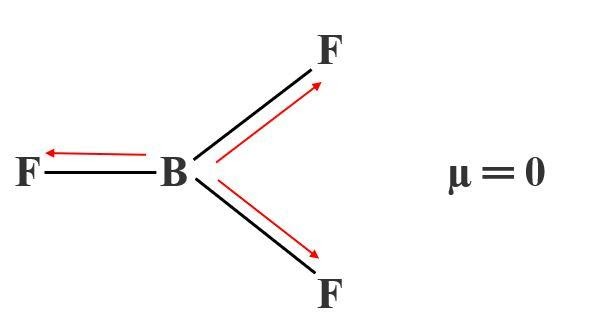
This happens in molecules with sufficiently symmetric shape. The mathematical definition of the dipole moment involves adding up the size of each charge in the molecule multiplied by the average distance that charge is from an arbitrary origin.Īny molecule where the overall centre of the positive charges and the overall centre of the negative charges coincide will have zero dipole moment. The dipole moment is a measure of the unevenness, or lack of symmetry, of the charge distribution in a molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
